Authors: Dr Pedro Aragon-Fernández1, A Prof Valdemaras Petrosius1, Greta Briedyte2, Dr Jarrod J. Sandow2, A Prof. Erwin M. Schoof1
Affiliations: 1Department of Biotechnology and Biomedicine, Cell Diversity Lab, Technical University of Denmark, 2IonOpticks, Collingwood, Australia.
Keywords: Aurora Rapid column, HeatSync, column heater, heater controller, Orbitrap, Astral, mass spectrometer, Vanquish Neo, UHPLC system, FAIMS, nano flow, 144 SPD, large cohort, clinical, high-throughput, HeLa tryptic digest, single-cell, QC runs, Spectronaut 20.2, retention time stability, backpressure monitoring.
ABSTRACT
Large-scale proteomics studies demand dependable LC-MS performance capable of sustaining high-throughput and reproducible results across thousands of injections. As experimental scales increase, particularly in single-cell proteomics and longitudinal studies, the ability of chromatographic systems to deliver consistent results over extended operation becomes critical. Ensuring column stability throughout prolonged use is therefore essential for maintaining both sensitivity and accuracy in quantitative data, as even minor shifts in performance can compromise downstream analysis and biological interpretation.
INTRODUCTION
The Aurora® Rapid™ 8 cm × 75 µm XT column, used in combination with a PepMap trap column, was employed to analyse a 4000+ sample single-cell proteomics dataset on a Thermo Scientific Vanquish Neo UHPLC system coupled to a Thermo Scientific Orbitrap Astral mass spectrometer. To ensure that the system was performing to a high standard, regular quality control (QC) assessments of LC-MS performance and reproducibility were carried out via the injection of HeLa tryptic peptide digests using the 144 SPD method. Between each QC run, approximately 60 single-cell runs were analysed, enabling real-world evaluation of column lifetime and long-term performance stability under demanding analytical conditions.
Experiment overview

High-throughput analysis of peptides and proteins
A HeLa tryptic digest (50 pg) was separated on an Aurora® Rapid™ 8 cm × 75 µm XT column using a 144 SPD method on the Thermo Scientific Vanquish Neo LC coupled to a Thermo Scientific Orbitrap Astral mass spectrometer, consistently identifying over 3,000 unique protein groups (Figure 1) and 15,000 unique peptides (Figure 2) per run.
Unique protein groups per run
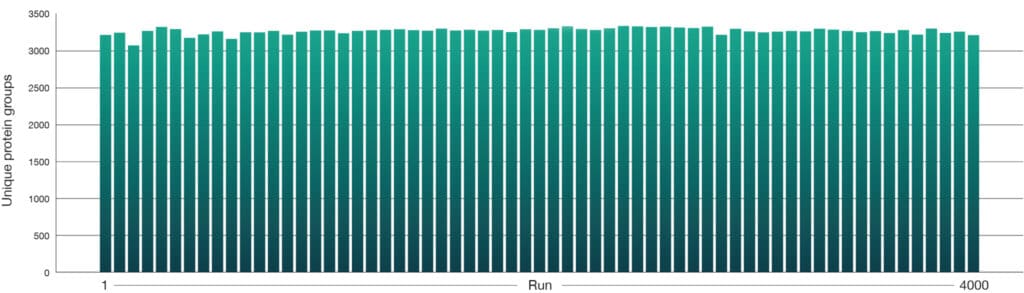
Figure 1: Unique protein group identifications from routine HeLa QC runs (50 pg) collected throughout a 4000‑sample experiment dataset on Aurora® Rapid™ 8 cm × 75 µm column with a PepMap trap column using a 144 SPD gradient. Samples were run on a Thermo ScientificVanquish Neo LC and Thermo Scientific Orbitrap Astral mass spectrometer. Raw data were analysed in Spectronaut version 20.2. A total of 63 QC runs were performed across the 4000 sample cohort.
Unique peptides per run
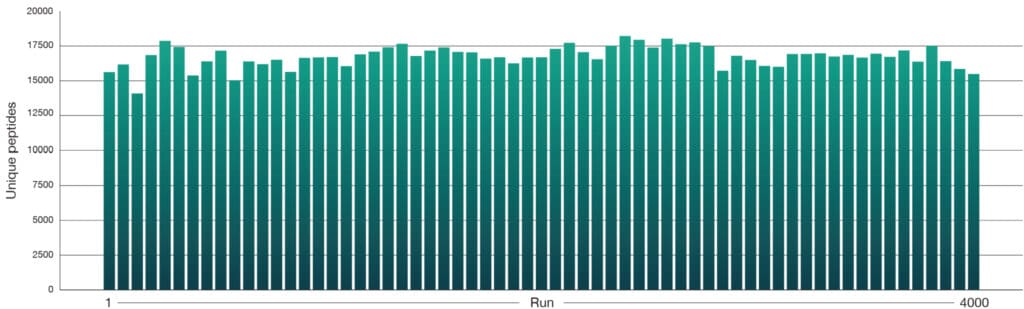
Figure 2: Unique peptide identifications from routine HeLa QC runs (50 pg) collected throughout a 4000‑sample experiment dataset on Aurora® Rapid™ 8 cm × 75 µm column with a PepMap trap column using a 144 SPD gradient. Samples were run on a Thermo ScientificVanquish Neo LC and Thermo Scientific Orbitrap Astral mass spectrometer. Raw data were analysed in Spectronaut version 20.2. A total of 63 QC runs were performed across the 4000 sample cohort.
Stable back-pressure over 4000 injections
By extracting and monitoring backpressure values during the analysis of QC samples, consistency was maintained across all 4,000 injections, demonstrating exceptional column stability and robustness over extended use (Figure 3).
Back-pressure value at 5 minutes
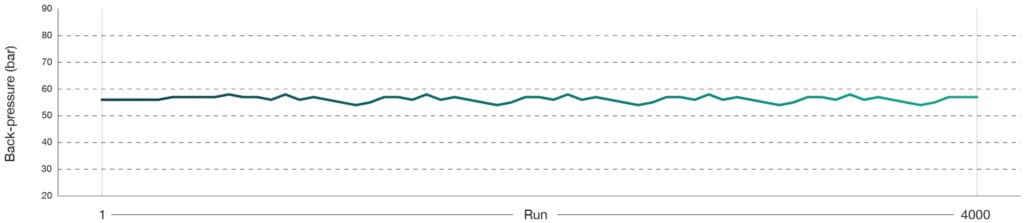
Figure 3: Back‑pressure values at 5 minutes from routine HeLa QC runs (50 pg) collected throughout a 4000‑sample experiment dataset on an Aurora® Rapid™ 8 cm × 75 μm column with a PepMap trap column using a 144 SPD gradient. The LC system operated at a flow rate of 0.1 μL/min with the heater controller set to 50 °C. Samples were run on a Thermo Scientific Vanquish Neo LC and Thermo Scientific Orbitrap Astral mass spectrometer. A total of 63 QC runs were performed across the 4000 sample cohort.
Stable retention times ensure confidence in results
This plot illustrates retention‑time stability over 4000 consecutive injections on a single Aurora® Rapid™ 8 cm × 75 µm column (Figure 4). Retention times remained highly consistent throughout the entire experiment, with minimal drift observed, demonstrating outstanding chromatographic reproducibility essential for large cohort projects.
Peptide retention time consistency (peptides found in all runs)
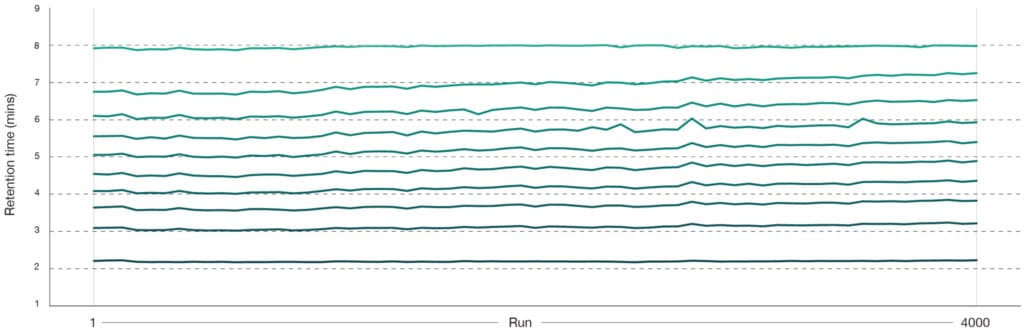
Figure 4: Peptide retention time stability from routine HeLa QC runs (50 pg) collected throughout a 4000‑sample experiment dataset on an Aurora® Rapid™ 8 cm × 75 μm column with a PepMap trap column using a 144 SPD gradient. Ten representative peptides were monitored, and their retention time were plotted across the 4000 injections. Samples were run on a Thermo Scientific Vanquish Neo LC and Thermo Scientific Orbitrap Astral mass spectrometer. Raw data were analysed in Spectronaut version 20.2. A total of 63 QC runs were performed across the 4000 sample cohort.
Excellent reproducibility across QC injections
A Pearson correlation matrix of all quantified protein intensities demonstrated exceptional reproducibility, with a mean Pearson correlation coefficient exceeding 0.99 across all 63 QC injections, indicating highly consistent protein quantification throughout the study (Figure 5).
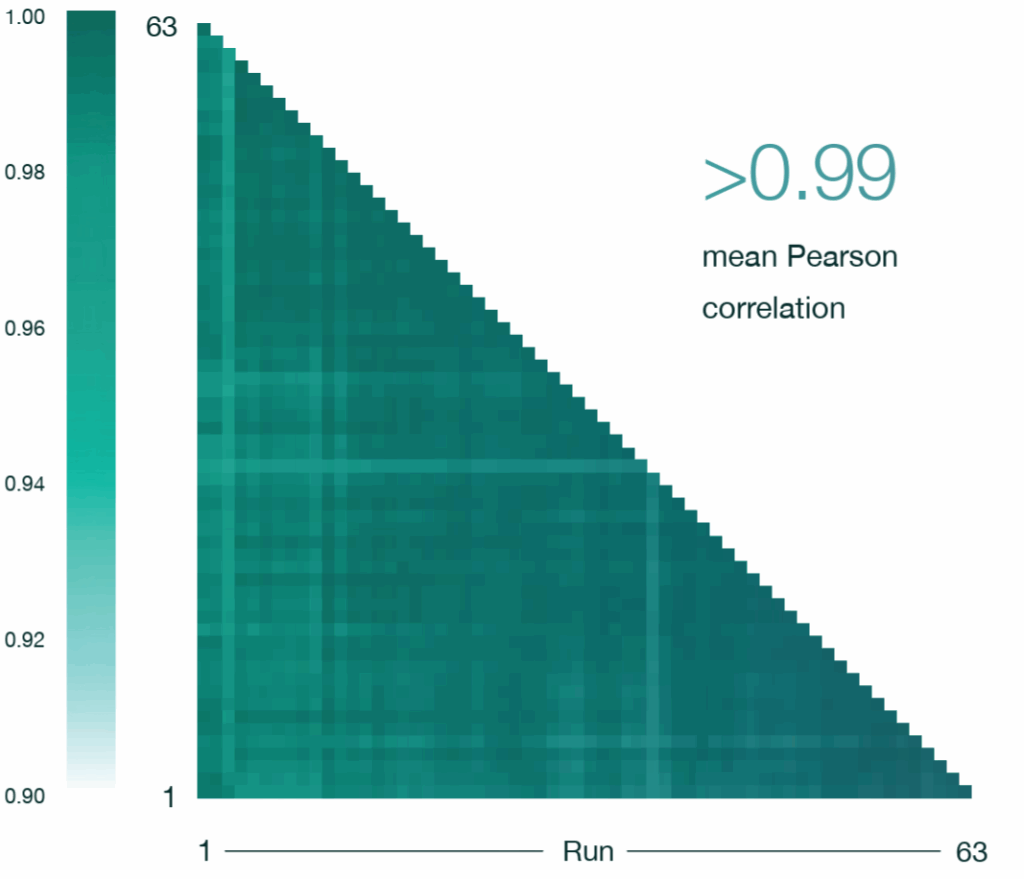
Figure 5: A Pearson correlation matrix of all quantified protein intensities from the 63 HeLa QC runs (50 pg) collected throughout the 4000‑sample experiment dataset on an Aurora® Rapid™ 8 cm × 75 μm column with a PepMap trap column using a 144 SPD gradient. Samples were run on a Thermo Scientific Vanquish Neo LC and Thermo Scientific Orbitrap Astral mass spectrometer. Raw data were analysed in Spectronaut version 20.2.
METHODS
LC-MS Analysis
Chromatographic separation of peptides derived from single cells and QC samples was performed on a Thermo Vanquish Neo UHPLC system connected to an 8 cm x 75 μm Aurora Rapid column heated to 50 °C. The autosampler and injection valves were configured to reverse flush trap-and-elute using a PepMap Neo Trap Cartridge (ThermoFisher Scientific). An active gradient of 8 minutes was applied, where the percentage of solvent B (80 % ACN and 0.1 % FA in H2O) was initially increased from 12 % to 30 % in the first minute, the nominal flow rate set at 500 nL/min. This was followed by a 10 % increase in solvent B during the second minute and a final elution step at 99 % solvent B with a reduced flow rate of 100 nL/min for the remainder of the gradient. All single-cell samples were injected at random from 384-well plates.
MS Parameters
For single-cell and QC sample analysis, MS1 spectra were acquired at an Orbitrap resolution of 120,000 with the same scan range of 400–800 m/z, an AGC target of 500 %, and maximum injection time of 100 ms. DIA of MS2 spectra was carried out on the ThermoFisher Astral using loop control with 13 spectra per cycle and isolation windows set to 20 m/z. The maximum injection time was set to 20 ms, and HCD was again applied at 25%. Fragment ions were detected using thesame scan range of 150–2000 m/z.
Data Processing
Raw data were analysed in Spectronaut version 20.2.
CONCLUSION
Together, these findings demonstrate that the Aurora® Rapid™ 8 cm × 75 µm column delivers exceptional robustness and reproducibility under extended use. Over the course of 4000 injections, the column consistently maintained stable backpressures and retention times, and sustained sensitivity, confirming its suitability for large‑scale, high‑throughput, and longitudinal proteomic studies.
ACKNOWLEDGEMENTS
We thank Associate Professor Erwin M. Schoof and his team at the Technical University of Denmark, where all data was acquired, and express our gratitude to Dr. Pedro A. Fernández for his exceptional efforts in preparing and running all LC-MS samples, overseeing the experimental execution, and providing invaluable assistance in preparing this application note.
Narrow peak widths
The plot highlights the average full width at half maximum (FWHM) values across all QC runs, showcasing consistently sharp and narrow peaks (Figure 6). This outstanding stability reflects the column’s superior reproducibility, delivering the reliable performance researchers need for confident, high‑throughput proteomic workflows.
Median FWHM per run
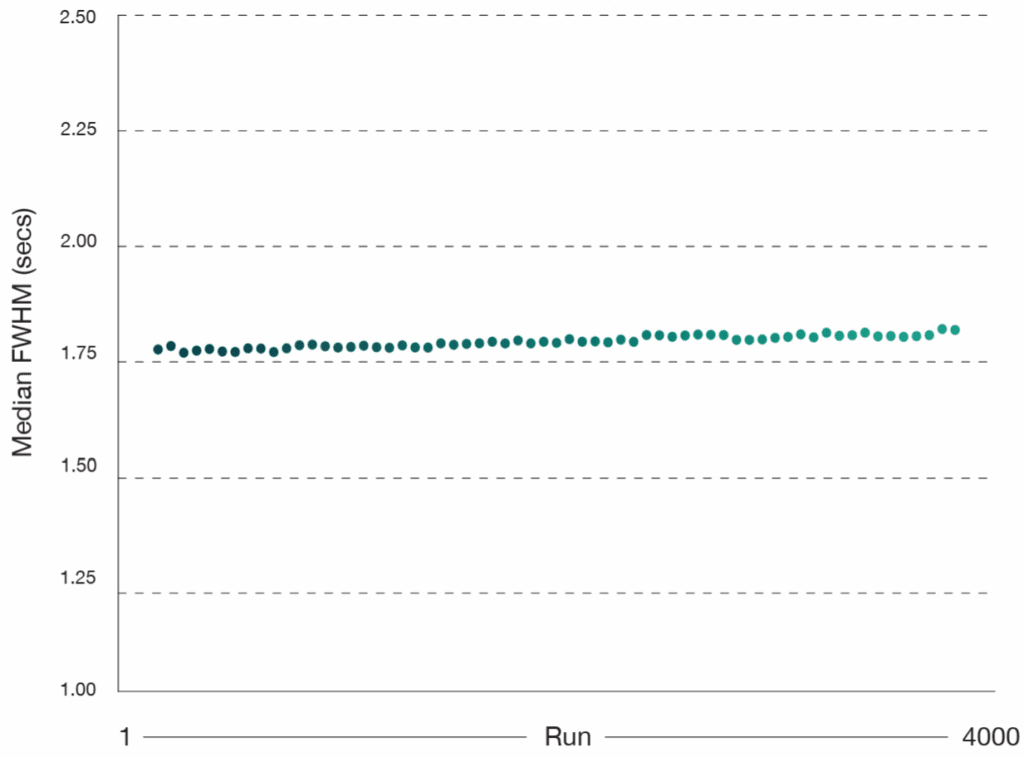
Figure 6: FWHM values from routine HeLa QC runs (50 pg) collected throughout a 4000‑sample experiment dataset on an Aurora® Rapid™ 8 cm × 75 μm column with a PepMap trap column using a 144 SPD gradient. Samples were run on a Thermo Scientific Vanquish Neo LC and Thermo Scientific Orbitrap Astral mass spectrometer. Raw data were analysed in Spectronaut version 20.2. A total of 63 QC runs were performed across the 4000 sample cohort.



FURTHER READING
For further resources and technical support, visit our Help Centre at helpcentre.ionoptickscopy.com. To view other application notes, read the latest publications featuring Aurora Series columns, or view the full range of IonOpticks products, visit our website at www.ionoptickscopy.com
About IonOpticks
IonOpticks produces high-performance chromatography solutions for the global research community. We specialise in the development and manufacture of columns for analytical applications in liquid chromatography with mass spectrometry (LC-MS) and high-end proteomics. Our highly reproducible methods provide a unique ability to enhance the sensitivity of mass spectrometry sample analysis, enabling scientists and clinicians to discover more from their samples. Our team are experts in a broad array of LC-MS platform technologies and are driven by the need to improve chromatographic performance in order to achieve data quality and deep proteome coverage on a whole new scale.