NanoShield™ 5×100 NZ C18 trap column 3-Pack

NanoShield™ 5×100 NZ C18 trap column 3-Pack
5 cm x 100 μm C18 UHPLC trap column with 2 x nanoZero® fittings compatible with nanoViper™
| SPD | 10 - 300 |
|---|---|
| Gradients | Use standard Aurora Series® analytical gradients for elution |
| Mode | Reversed-flush |
Available on back-order
Chromatographic trap columns are essential in proteomic UHPLC–MS workflows, improving analyte focusing and enabling rapid sample loading with in-line sample clean-up to protect analytical columns from blockages and contamination while reducing ion suppression. These benefits extend column lifetime and reduce maintenance, supporting robust, high-throughput, and reproducible proteomic analyses. Despite these advantages, trap columns traditionally sacrifice peptide and protein identifications compared to direct injection. The NanoShield™ C18 5 cm × 100 μm trap column eliminates this trade-off – delivering direct injection-level performance; providing superior peptide identifications, peak quality, and hydrophilic peptide coverage compared to other traps.
Product benefits
- Next to no peptide ID loss
- Extends column life, reduces maintenance costs
- Improved reproducibility across thousands of injections
- Enables hundreds of samples per day
Ideal for
- Fast sample loading
- Reversed-flush workflows
- Bottom-up discovery workflows
A perfectly paired trap for Aurora Series columns.
A 100 ng HeLa tryptic digest was analysed using a reverse-flush trap-elute method compared to direct injection across two workflows: a 37-minute gradient on the Aurora® Ultimate™ CSI (25 cm x 75 μm) column and a 14-minute gradient on the Aurora® Rapid™ CSI (8 cm × 75 μm) column, both analysed on a Bruker nanoElute 2 UHPLC coupled to a Bruker timsTOF HT mass spectrometer.
The NanoShield™ trap column (5 cm x 100 μm, 3 μm C18 particles) demonstrated near-direct injection performance across all metrics while substantially outperforming a competitor C18 trap cartridge (5 mm x 300 μm, 5 μm C18 particles). NanoShield™ showed less than 1% reduction in protein identifications and a 1–3% reduction in precursor identifications compared to direct injection across both columns.
Peak quality metrics remained similarly robust, with marginal increases in FWHM and minimal changes to peak shape ratios compared to direct injection, while maintaining significantly superior performance relative to the competitor trap.
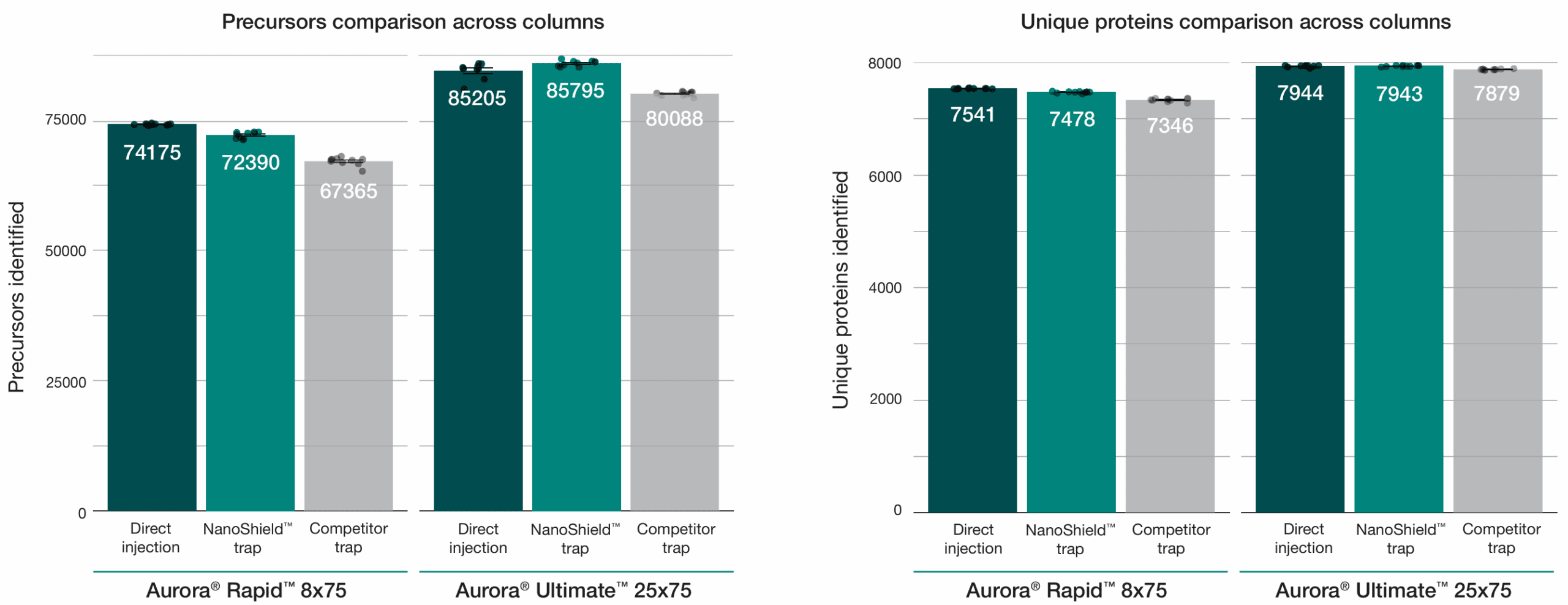
A 100 ng HeLa tryptic digest was analysed across two workflows: a 37-minute gradient on the Aurora® Ultimate™ CSI (25 cm x 75 μm) column and a 14-minute gradient on the Aurora® Rapid™ CSI (8 cm × 75 μm) column, both analysed on a Bruker nanoElute 2 UHPLC coupled to a Bruker timsTOF HT mass spectrometer. NanoShield™ maintains near-direct injection identification rates across column formats. Precursor and unique protein identifications from 100 ng HeLa digest comparing direct injection, the 5cm NanoShield™ trap column, and a competitor C18 trap cartridge on an Aurora® Rapid™ 8 cm × 75 μm column (14-min gradient) and a Aurora® Ultimate™ 25 cm x 75 μm column (37-min gradient). Data analysed with Spectronaut 20.2. Each dot represents the analysis of an independent sample injection.
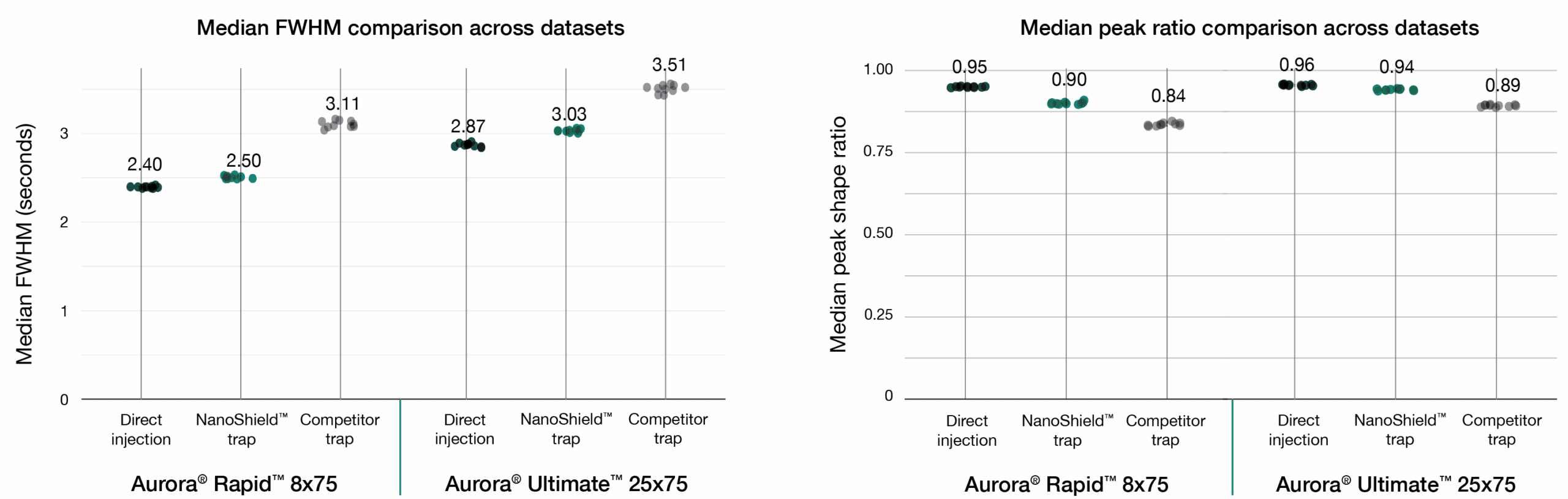
A 100 ng HeLa tryptic digest was analysed across two workflows: a 37-minute gradient on the Aurora® Ultimate™ CSI (25 cm x 75 μm) column and a 14-minute gradient on the Aurora® Rapid™ CSI (8 cm × 75 μm) column, both analysed on a Bruker nanoElute 2 UHPLC coupled to a Bruker timsTOF HT mass spectrometer. NanoShield™ preserves peak quality with minimal performance loss. FWHM and peak shape ratio comparisons for direct injection, NanoShield™ trap, and a competitor C18 trap cartridge on an Aurora® Rapid™ 8 cm × 75 μm column (14-min gradient) and a Aurora® Ultimate™ 25 cm x 75 μm column (37-min gradient). Data analysed with Spectronaut 20.2. Each dot represents the analysis of an independent sample injection.
Superior peptide rentention.
One disadvantage of using traditional trap columns is the risk of losing hydrophilic peptides during sample loading. Early-eluting peptides such as post-translationally modified peptides (e.g., phosphopeptides and glycopeptides), which are typically more hydrophilic, can pass through traditional traps during the sample loading and clean-up steps, creating gaps in proteome coverage and reducing overall identifications. This hydrophilic peptide loss compromises the comprehensive analysis that direct injection workflows provide. The NanoShield™ C18 trap column eliminates this barrier, delivering direct injection-level proteome coverage and identifications as demonstrated above.
To quantify peptide retention across the gradient, a reference set of 100 peptides equally distributed by retention time was selected from the Aurora® Ultimate™ 25 cm × 75 μm direct injection analysis. Only high-abundance peptides (top 25% intensity) were selected to ensure reliable detection. These peptides were then tracked across both the NanoShield™ and a competitor C18 trap cartridge. The competitor C18 trap cartridge failed to retain the majority of early-eluting peptides, while the NanoShield™ successfully captured all 100 peptides observed in direct injection (Figure 3). Additionally, the NanoShield™ demonstrated stable retention times across multiple injections, ensuring reproducible peptide elution profiles
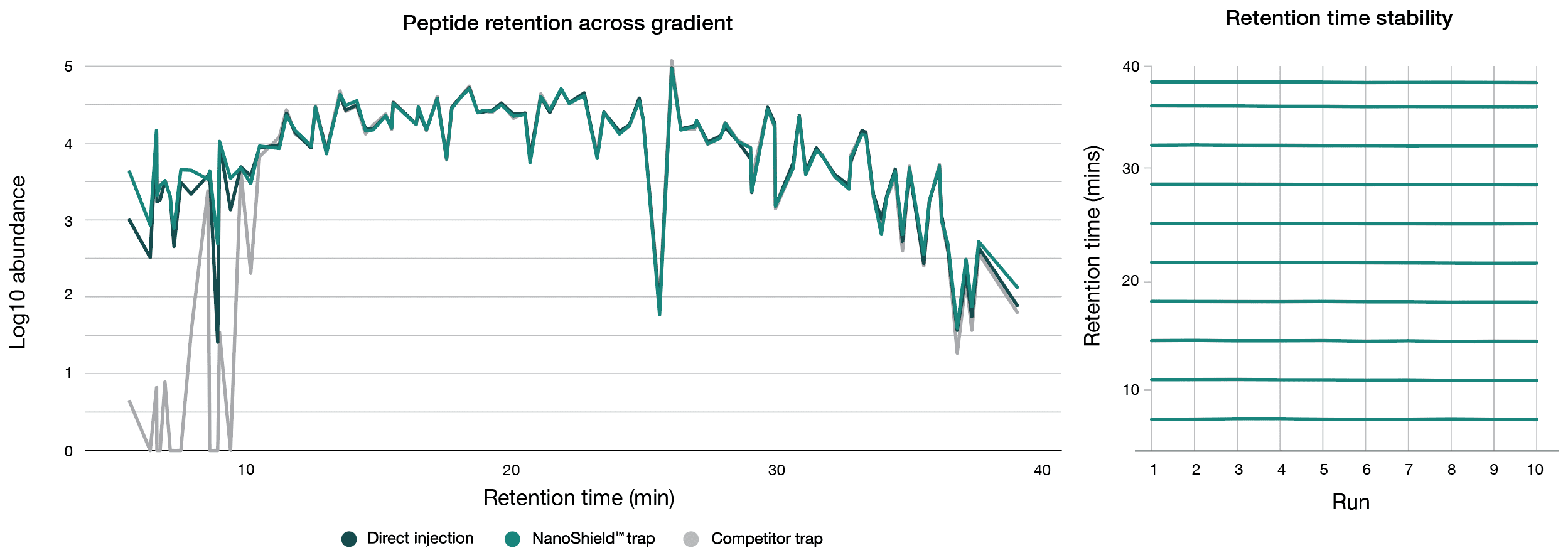
A 100 ng HeLa tryptic digest was analysed across a 37-minute gradient on the Aurora® Ultimate™ CSI (25 cm x 75 μm) column on a Bruker nanoElute 2 UHPLC coupled to a Bruker timsTOF HT mass spectrometer. (Left) NanoShield™ provides superior peptide retention across the chromatographic gradient. Comparing direct injection against both NanoShield™ and a competitor C18 trap cartridge coupled to an Aurora® Ultimate™ (25 cm × 75 μm) column, NanoShield™ fully retains early-eluting peptides, which are lost with the competitor C18 trap cartridge. (Right) Retention time stability with NanoShield™ trap column for 10 peptides across multiple injections on an Aurora® Ultimate™ 25 cm x 75 μm column.
Narrow peak widths
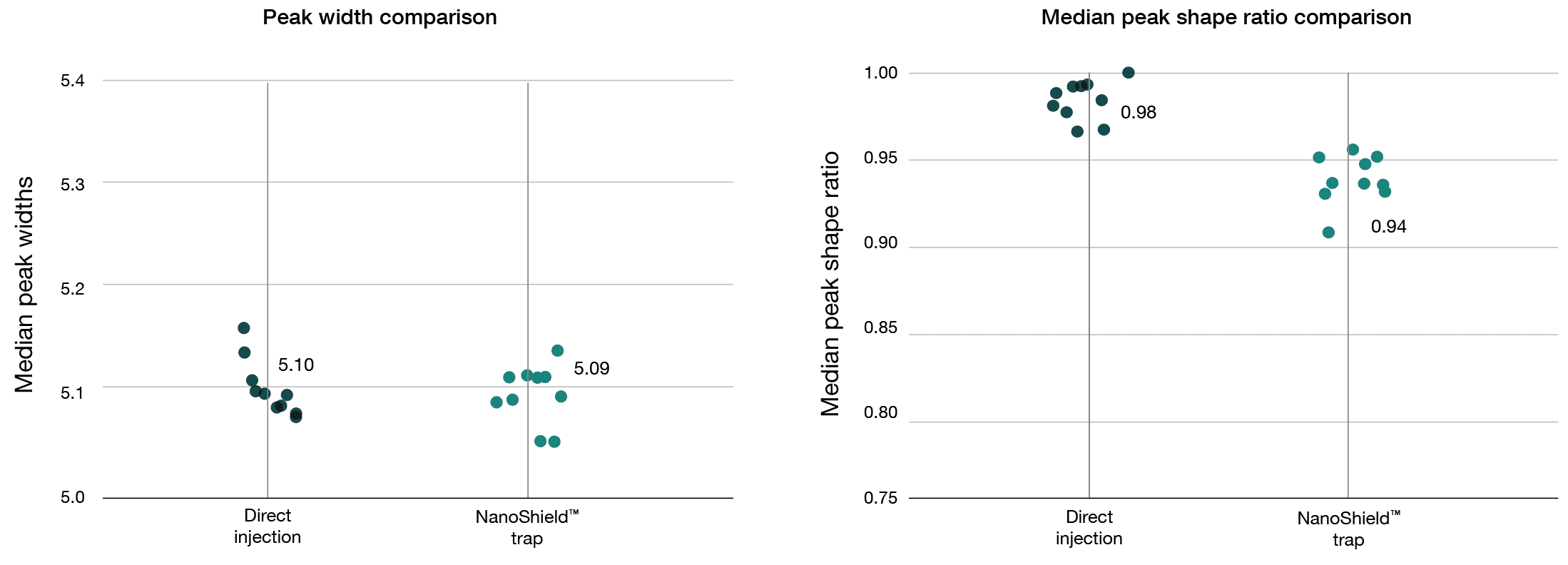
A 100 ng HeLa tryptic digest was analysed using a reverse-flush trap-elute method and compared to direct injection on a 40-minute gradient, employing an Aurora® Ultimate™ XT 25 cm × 75 μm on an UltiMate 3000 UHPLC coupled to a Thermo Scientific Orbitrap Fusion Lumos mass spectrometer. NanoShield™ preserves peak quality with minimal performance loss. FWHM and peak shape ratio comparisons for direct injection, NanoShield™ and an Aurora Ultimate XT 25cm x 75 μm column (40-min gradient). Raw data was analysed using FragPipe (version 22.0) and MaxQuant (version 2.6.7.0). Each dot represents the analysis of an independent sample injection.
| Column format | Trap/guard column |
|---|---|
| Column type | Reversed-phase |
| Trap column orientation | Reverse flush |
| For use with | UHPLC |
| Length | 5 cm |
| Inner Diameter | 100 µm |
| Pore size | 120 Å |
| Pressure | >1700 bar |
| Particle size | 3 µm |
| pH stability | 1–8 |
| Stationary phase | C18 |
| Suggested loading capacity | 200 ng |
| Max loading capacity | 1 µg |
| Suggested loading pressure | 800 bar |