Photo modified from kues1 on Freepik.
Metabolic disorders present a significant global health challenge, with diabetes cases in the US projected to increase 165% by 2050. While β-cell dedifferentiation and loss of identity are known contributors to diabetes pathogenesis, much remains unknown about the molecular diversity and biology of pancreatic islets, particularly regarding rare endocrine states and cellular plasticity. Single-cell transcriptomics can reveal significant endocrine heterogeneity, however spatial proteomic profiling at deep coverage has lagged behind due to technical limitations.
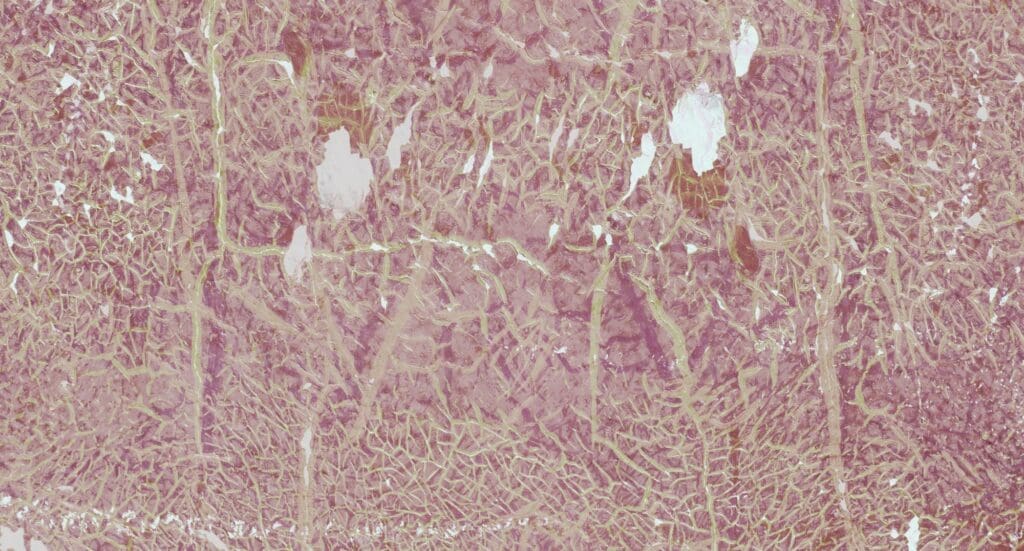
A new study led by Mardamshina et al. from the Lundberg Laboratory at Stanford University introduces multiplexed Deep Visual Proteomics (mxDVP), an end-to-end pipeline integrating high-plex microscopy with ultra-sensitive mass spectrometry to enable spatially resolved proteomic profiling. Using laser microdissection and AI-guided annotation, they precisely isolated and analysed islet cell subpopulations from multiplexed tissue sections across multiple donors. Proteomic analyses were then conducted using Aurora Elite XT 15×75 C18 UHPLC columns with an Evosep One LC system running a Whisper 40 SPD method coupled to an Orbitrap Astral with EASY-Spray source and operated in DIA mode with FAIMS, or a Whisper 20 SPD method coupled to a Bruker timsTOF SCP with a CaptiveSpray source and operated in dia-PASEF mode. This approach revealed ~6,000 unique proteins and distinct proteomic signatures across classical islet cell types.
This exploratory study identified and characterised 12 endocrine subtypes among 38,753 islet cells, including rare polyhormonal populations that are typically missed by conventional single-cell sequencing approaches. Pathway analysis revealed cell type-specific enrichments: β-cells showed upregulated metabolic pathways, while α-cells were enriched for hormone secretion functions. It was also demonstrated that larger islets predominantly composed of classical endocrine cells had reduced vascularisation, whereas smaller islets containing more polyhormonal cells were better vascularised. Notably, polyhormonal cells expressed transcription factors linked to progenitor states, suggesting a potential role in islet regeneration or plasticity.
By combining high-plex antibody panels, refined image analysis, and deep proteomic profiling despite minimal tissue requirements, the mxDVP approach establishes a potent framework for studying tissue heterogeneity across distinct physiological and pathological contexts, and uncovers molecular signatures of rare and transitional states. The crucial insights into islet cell fate dynamics in this study may inform future therapeutic strategies for diabetes and other metabolic disorders.
Publication
bioRxiv
Authors
Mariya Mardamshina, Nicolai Dorka, Marvin Thielert, Frida Björklund, Frederic Ballllosera Navarro, Anna Martinez Casals, Ferenc Kovacs, Jocelyn E. Manning Fox, Peter Horvath, Patrick E. MacDonald, Matthias Mann, & Emma Lundberg;
Title