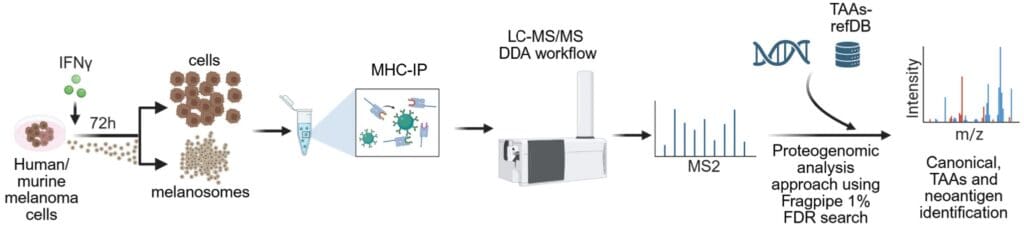
Experimental scheme of the proteogenomic immunopeptidome analysis workflow. From Chemla et al., 2025. “HLA export by melanoma cells decoys cytotoxic T cells to promote immune evasion.“ Cell (2025). doi: 10.1016/j.cell.2025.11.020; Licensed under the terms of the Creative Commons CC-BY 4.0 license.
Melanoma remains highly lethal, with approximately 50% of patients resistant to immunotherapy despite the presence of tumour antigens recognised by tumour-infiltrating lymphocytes (TILs). The mechanisms underlying this immune evasion have been incompletely understood. Chemla et al. investigated whether melanosomes, large extracellular vesicles (200-500 nm) unique to melanoma cells, directly modulate T cell function and contribute to immune escape.
The team from the Levy lab used a multi-faceted experimental approach combining mass spectrometry-based proteomics, immunopeptidomics, single-cell RNA sequencing, and T cell receptor sequencing. For proteomic analysis, melanosomes and exosomes were isolated from human and mouse melanoma cultures and analysed using LC-MS/MS. A nanoElute 2 was coupled with an Aurora® Ultimate™ 25×75 CSI C18 UHPLC column via CaptiveSpray source to a timsTOF Ultra operating in DDA-PASEF mode. Additional experiments employed Q Exactive HF-X, Q Exactive HF, and Exploris 480 mass spectrometers with various LC systems and column setups.
This exploratory study revealed that melanoma-derived melanosomes are significantly enriched in immune-related proteins compared with exosomes, particularly those involved in MHC class I antigen presentation. Immunopeptidomic analysis identified 25 tumour-associated antigens and three neoantigens presented by melanosomes, with melanosomes showing statistically significant enrichment in tumour-associated antigen presentation compared with melanoma cells. Leveraging single-cell T cell receptor sequencing, the researchers demonstrated that melanosomes carry HLA molecules loaded with immunogenic peptides that directly engage CD8+ T cell receptors, competing with tumour cells for T cell recognition. This interaction delivers incomplete activation signals, reduces mitochondrial activity, and induces T cell apoptosis, effectively acting as decoys that shield melanoma cells from immune attack.
This discovery identifies melanoma-derived melanosomes as active mediators of immune evasion and potential therapeutic targets, suggesting that blocking melanosome secretion or excluding melanosome-reactive TILs from adoptive cell therapy could enhance immunotherapy responses in melanoma patients.
Publication
Cell
Authors
Yoav Chemla, Orit Itzhaki, Stav Melamed, Chen Weller, Yuval Sade, Paulee Manich, Keren Reshef, Nicolas Xenidis, Avishai Maliah, Gilad Levy, Roma Parikh, Osnat Bartok, Opal Levy, Itay Tal, Gal Aziel, Abraham Nissani, Sharon Yunger, Daniela Likonen, Vitaly Kliminsky, Tamar Golan, Coralie Capron, Valentina Ace, Ronen Levy, Diana Rasoulouniriana, Zohar Eyal, Yuval Barzilay, Roi Balaban, Aseel Khateeb, Rami Khosravi, Amir Grau, Tamar Ziv, Polina Greenberg, Dvir Netanely, Hananya Vaknin, Xunwei Wu, Yael Amitay, Ronen Brenner, Julia María Martínez Gómez, Dov Hershkovitz, Tal Yardeni, Valentina Zemser-Werner, Oren Kobiler, Yael Friedmann, David Bassan, Ron Shamir, Lea Eisenbach, Nadine Santana-Magal, Michael Milyavsky, Galit Eisenberg, Leeat Keren, Merav Cohen, Dvir Gur, Boaz Barak, Michal Lotem, David Sprinzak, Shoshana Greenberger, David Fisher, Michal J. Besser, Mehdi Khaled, Pierre Close, Ronnie Shapira, Sebastien Apcher, Asaf Madi, Mitchell P. Levesque, Francessca Rapino, Yaron Carmi, Shivang Parikh, Yardena Samuels, & Carmit Levy;
Title
HLA export by melanoma cells decoys cytotoxic T cells to promote immune evasion

